An Everyday DNA blog article
Written by: Sarah Sharman, PhD, Science writer
Illustrated by: Cathleen Shaw
Here in the south, we spend many spring days glued to the weather forecast seeking early warning about approaching storms. Meteorologists study radars, atmospheric stability, temperature, humidity, and other factors to predict where tornadoes may form. Then they release a warning to people in areas that could be affected, giving them time to seek shelter from the storm.
Similarly, there are many warning signs in our bodies that alert doctors to the presence of disease or illness. These signs, called biomarkers, have become the focus of an increasing number of studies over the past few decades. Let’s learn what biomarkers are and how they are useful in biomedical research and healthcare.
What is a biomarker?
Biomarkers, short for biological markers, are characteristics that are measured to determine health status, the presence and progression of disease, or the effects of a treatment. Anything that helps in the identification of disease can serve as a biomarker. Common types of biomarkers include specific cells, molecules, genes, gene products, enzymes, hormones, or complex organ functions.
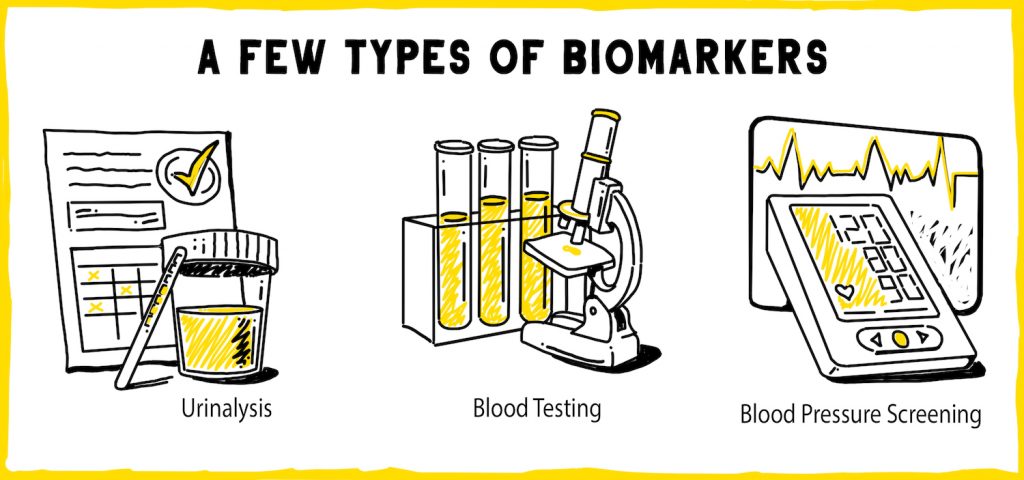
 Many biomarkers, like heart rate, blood pressure, urinalysis, and x-ray findings, are commonly used in the clinic and well-researched. You have likely had some of these measured during a routine check-up with your doctor. Advances in molecular biology and genomic sequencing technology have led to the emergence of new types of biomarkers, like genes and nucleic acids, that are still being characterized and integrated into clinical practice.
Many biomarkers, like heart rate, blood pressure, urinalysis, and x-ray findings, are commonly used in the clinic and well-researched. You have likely had some of these measured during a routine check-up with your doctor. Advances in molecular biology and genomic sequencing technology have led to the emergence of new types of biomarkers, like genes and nucleic acids, that are still being characterized and integrated into clinical practice.
The table below outlines some of the more common types of biomarkers used in the research and medical fields today.
Biomarkers for use in biomedical research and healthcare should be safe, cost-effective, easy to measure, and be able to accurately differentiate between health and disease states. The ideal sample material for a biomarker test should be easy to obtain and as non-invasive as possible. This could include a blood sample taken by a doctor, a urine or saliva sample, or a drop of blood like those diabetes patients extract from their own fingertips for regular blood-sugar monitoring.
What are some utilities of biomarkers?
Biomarkers have many useful applications in health care, including disease prevention and detection, determination of individual disease risk, and disease monitoring. They are being used more frequently in clinical trials to measure safety, toxicity, and efficacy of emerging therapeutics.
The table below outlines common uses for biomarkers in the medical and clinical trials fields.
The use of validated biomarkers in clinical care has become commonplace in the diagnosis and management of many diseases such as cardiovascular disease, infections, immunological and genetic disorders, and cancer. Biomarkers allow for better understanding of disease processes and the ways in which drugs work to combat disease.
Biomarkers are becoming more and more important for chronic disease diagnosis because they can confirm a difficult diagnosis or make a diagnosis that was not possible in the first place. Some chronic diseases, such as Alzheimer disease and rheumatoid arthritis, often begin with an early, symptom-free phase. Biomarkers can help to quickly and reliably identify individuals at high-risk of developing symptoms so that they can either be treated before, or shortly after, disease onset.
The oncology field has also benefited largely from the discovery and implementation of biomarkers into regular practice. They help doctors detect the presence of cancer, monitor the progression of cancer, predict the best treatment for the patient, and even determine who is at a greater risk of developing cancer.
Biomarker research
In basic and clinical research, biomarkers can be used to improve the efficiency and safety of existing therapeutic drugs, develop new therapeutics, and aid in the diagnosis of disease. New molecular biomarkers have the potential to personalize disease prevention and management, making the delivery of healthcare more precise, safe, and cost effective.
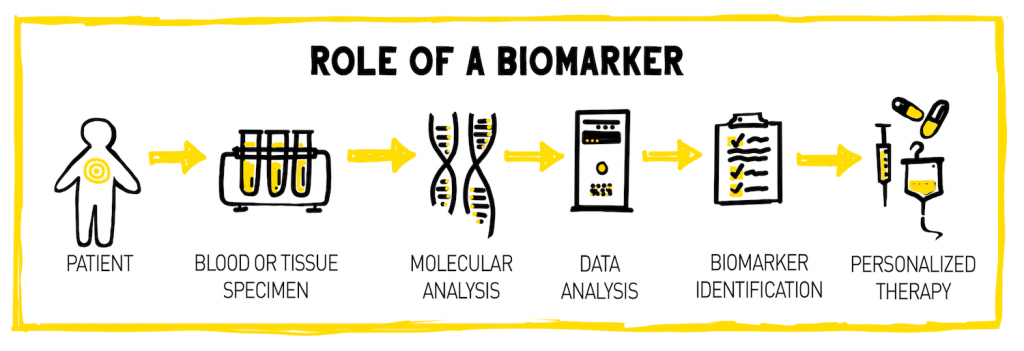 Scientists at the HudsonAlpha Institute for Biotechnology are part of this quest to discover new, clinically-relevant biomarkers for disease diagnosis and treatment. HudsonAlpha President, Science Director and M.A. Loya Chair in Genomics, Richard M. Myers, PhD, and his lab previously discovered circulating biomarkers of early colon lesions called colorectal adenoma that could eventually develop into colon cancer. The biomarkers were a type of plasma biomarker called cell-free nucleic acids which are DNA or RNA molecules that have been released into the blood by cells in response to cell damage, stress, or death.
Scientists at the HudsonAlpha Institute for Biotechnology are part of this quest to discover new, clinically-relevant biomarkers for disease diagnosis and treatment. HudsonAlpha President, Science Director and M.A. Loya Chair in Genomics, Richard M. Myers, PhD, and his lab previously discovered circulating biomarkers of early colon lesions called colorectal adenoma that could eventually develop into colon cancer. The biomarkers were a type of plasma biomarker called cell-free nucleic acids which are DNA or RNA molecules that have been released into the blood by cells in response to cell damage, stress, or death.
Using this same technology, the team, led by postdoctoral fellow Ben Henderson, PhD, and senior scientist Brian Roberts, aims to uncover biomarkers that can diagnose early-stage disease, track disease progression, and monitor therapeutic effectiveness in neurodevelopmental diseases like Alzheimer disease, Huntington disease and ALS. By comparing plasma from control samples to samples from patients diagnosed with a neurodegenerative disease, the team identifies small RNA molecules that are unique to patients with disease.
Through a small pilot study, the team has identified a potentially promising small RNA signature that could be used to diagnose Alzheimer disease earlier and track its progression. The team is also applying the same methodology to Huntington disease to find a unique small RNA signature that can help in its diagnosis. If this biomarker signature can be validated, they could become integrated into clinical care as a screen for people with a family history of neurodegenerative disease or people having undiagnosed neurological symptoms.
Biomarker screening tests for cancer predisposition are implemented in clinical care already. At HudsonAlpha, the Information Is Power initiative offers participants from five counties in North Alabama low-cost cancer predisposition screening. The screening test looks at several dozen genes associated with increased risk for breast, ovarian, colon, and many other cancers. The knowledge gained from the screening test can help patients and their doctors alter the patient’s care or identify cancer at an earlier stage.